Sharp has been demonstrating the effectiveness of Plasmacluster technology by working with independent third-party research organizations around the world.
For the first time in the world, a device, developed by Sharp Corporation, equipped with Plasmacluster technology, which exposed an airborne novel coronavirus (SARS-CoV-2) to Plasmacluster ions for approximately 30 seconds, has effectively reduced the infectious titer of the virus by more than 90%. This achievement was accomplished in collaboration with Professor Jiro Yasuda of the National Research Center for the Control and Prevention of Infectious Diseases/Institute of Tropical Medicine, Nagasaki University, Professor Asuka Nanbo (a board member of the Japanese Society for Virology) of the same institution, Professor Hironori Yoshiyama of the Department of Microbiology, Shimane University, Faculty of Medicine (also, a board member of the Japanese Society for Virology), and Nagasaki University, an internationally respected authority on infectious diseases research.
In 2004, Sharp demonstrated the effectiveness of Plasmacluster technology against feline (cat) coronavirus, a member of the Corona viridae family. In the following year of 2005, Sharp also demonstrated its effectiveness against the original SARS coronavirus (SARS- CoV), which caused the outbreak of 2002-2003 and is also genetically similar to the novel coronavirus (SARS-CoV-2). Now, Sharp has demonstrated its effectiveness against SARS-CoV-2 in airborne droplets.
Dr Jiro Yasuda, Professor of National Research Center for the Control and Prevention of Infectious Diseases, Nagasaki University said, “Disinfectants such as alcohol and detergents (surfactants) are well-known to be effective to reduce the risk of the virus on materials. However, for infection via aerosols (micro-droplets), there are few effective countermeasures such as a mask. Today, we demonstrated the effective inactivation of SARS-CoV-2 in airborne droplets by using Plasmacluster technology. We expect it would be useful to reduce the risk of infection in real spaces like offices, homes, medical facilities and vehicles too”.
Since 2000, Sharp has been demonstrating the effectiveness of Plasmacluster technology by working in collaboration with independent third-party research organizations around the world.
Research Details –
Overview of Verification Test
- Testing organization: National Research Center for the Control and Prevention of Infectious Diseases (CCPID)/Institute of Tropical Medicine, Nagasaki University
- Verification test apparatus: Virus testing device equipped with Plasmacluster technology
Plasmacluster ion concentration: Approx. 10 million/cm3 (in the vicinity of the Plasmacluster ion generator)
- Test space volume: Approx. 3 liter
- Control study: Comparison using the device described above without Plasmacluster ion generation
- Validation virus: Novel Coronavirus (SARS-CoV-2)
- Test method
-Pass the aerosolized virus through the test devise from the generator
-Recover the aerosolized virus after exposure to Plasma cluster ions
-Calculate the infectious virus titer of the recovered virus solution by a plaque assay*
* A standard assay to evaluate the number of infectious virus in the sample.
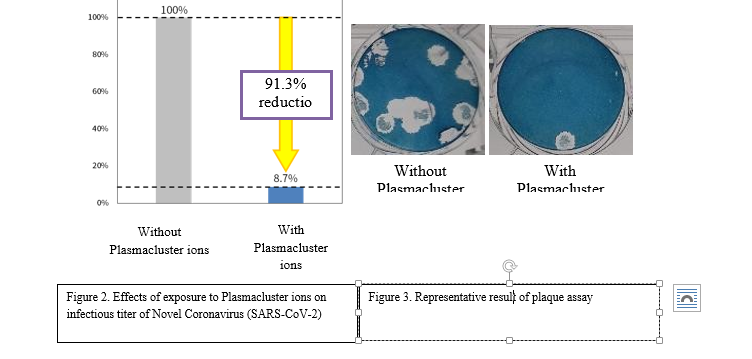
Results-
Table 1 Effect of Plasmacluster ions on infectious titer of novel coronavirus (SARS-CoV-2) suspended in the air.
| Without Plasmaclusterions | With Plasmacluster ions | Reduction | |
| Infectious virus titer (number of plaque) | 1.76 x 104 | WithoutPlasmacluster ionsWithPlasmacluster ions91.3%reduction1.54 x 103 | 91.3% |